 These vaccines often include an adjuvant to increase the body’s immune response. If you later become infected with the COVID-19 virus, the antibodies will fight the virus. Once your immune system recognizes the protein subunit, it creates antibodies and defensive white blood cells. Subunit vaccines include a part (protein) of the virus that best stimulates your immune system. 
These antibodies protect us from getting infected if the real virus enters our bodies. Once the spike protein is made, two things occur: (1) the cell breaks down the mRNA (instructions) and gets rid of them, and (2) the spike protein triggers an immune response inside of our bodies, which produces antibodies. The mRNA in the vaccine teaches our cells how to make copies of a spike protein. 
These vaccines use a newer technology where mRNA is wrapped in a coating (lipid nanoparticle) so it can enter our cells. What is the difference between COVID-19 mRNA, and protein subunit vaccines? FDA EUA Fact Sheet for Healthcare Professionals (ages 12+ years).Comirnaty Package Insert (ages 12+ years).FDA EUA Fact Sheet for Healthcare Professionals (ages 6 months – 11 years).Spikevax Package Insert (ages 12+ years).The US has two mRNA and one protein subunit COVID-19 vaccines approved for use. CDC Guidance on Interchangeability of COVID-19 VaccinesĬOVID Vaccine Products Which COVID-19 vaccines have been approved and/or authorized for use in the US?.AAP Pediatric COVID-19 Vaccine Dosing Quick Reference Guide.If not possible, use of another brand is acceptable. Whenever possible, use the same brand for all recommended doses in a primary series for children 6 months – 4 years of age. Children 6 months – 4 years of age who previously completed a primary series with one brand may switch to a different brand when receiving an updated dose, if the brand used for the primary series is not readily available on the day of vaccination. “Mix and match” dosing is allowed for children 5 years of age and older. Can you “mix and match” vaccine products for one child? See additional information in the AAP Pediatric COVID-19 Vaccine Dosing Guide. Which Moderna or Pfizer-BioNTech vaccine dose should a child receive if they will have a birthday between doses?Ī child should receive the age-appropriate vaccine product and dosage based on their age on the day of vaccination.For children who turn 4 to 5 years of age and are receiving Pfizer mRNA vaccine, if the 10 mcg dose is the second dose, administer 3-8 weeks after the first dose if it is the third dose, administer at least 8 weeks after the second dose. Also included is dosing information for children with moderately or severely immunocompromising conditions. This resource offers the dosing intervals for unvaccinated children and previously vaccinated children. Please the AAP Pediatric COVID-19 Vaccine Dosing Quick Reference Guide for dosing intervals. 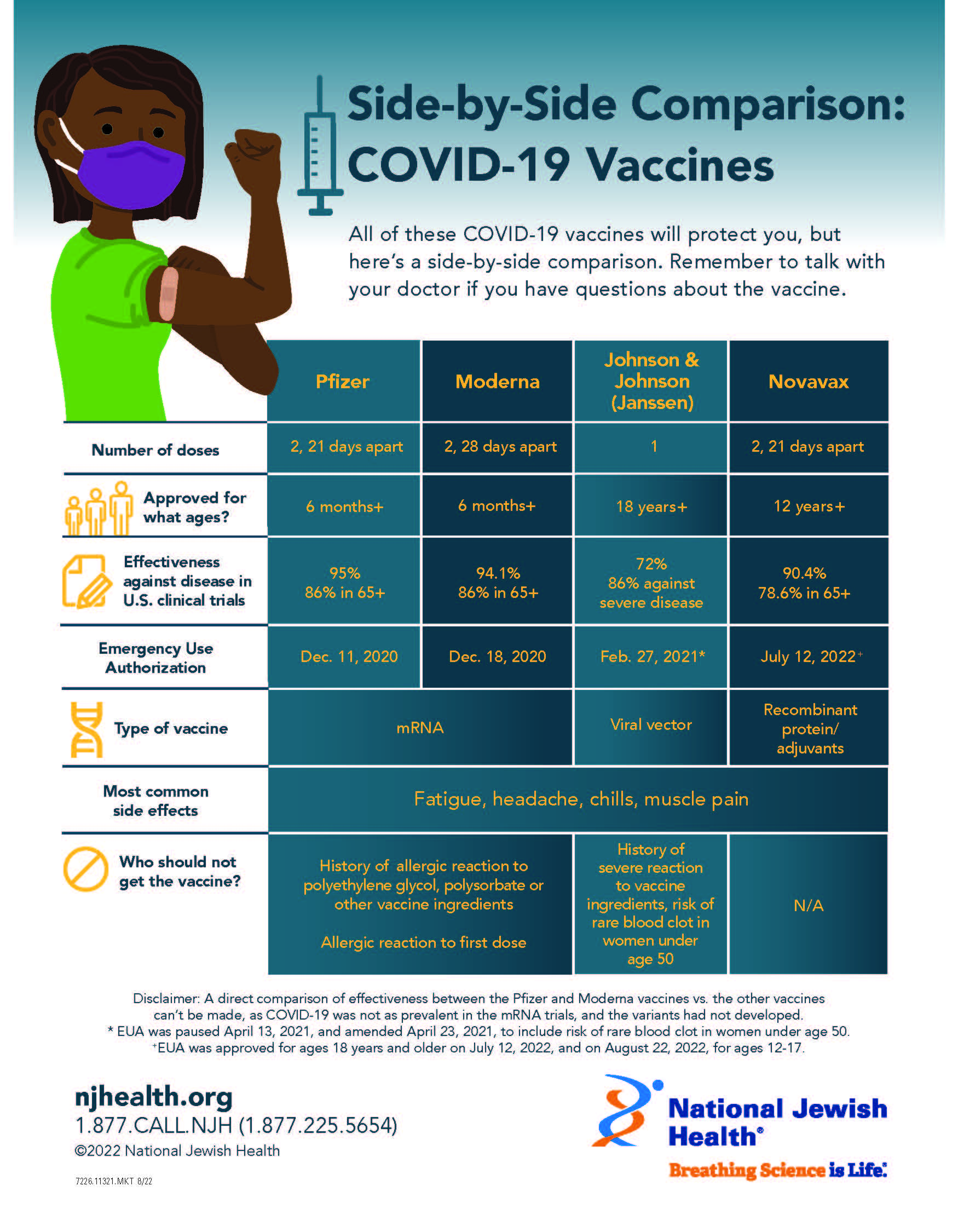
The FDA indicated that bivalent COVID vaccines are no longer authorized for use in the US on September 11, 2023.ĬOVID Vaccine Dosing What is the recommended interval between the doses of an mRNA COVID-19 vaccine series? How can I determine what my patient needs if they already received at least one monovalent COVID-19 dose If I haven’t yet received my supply of updated 2023-2024 vaccine and still have doses of bivalent vaccine, can I administer it? Individuals (6 months of age and older) who are moderately to severely immunocompromised may require additional doses. The updated dose should be given at least two months after any previous COVID vaccine dose.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
